Our Services and Facilities
The McLaughlin Research Institute is the closest biomedical research institution to more than two thirds of residents of Montana. We seek to bring opportunity for participation in biomedical research and education to those in urban and rural communities across the state.
Transgenic Facility
-
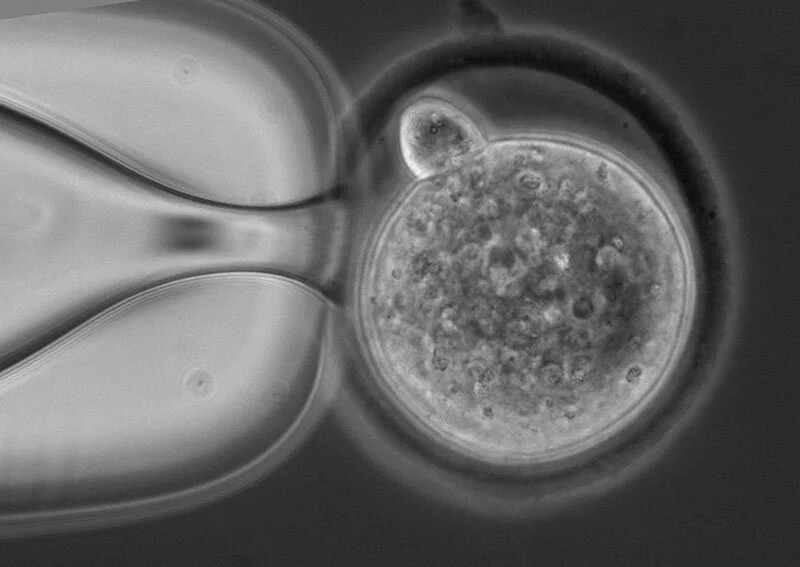
The McLaughlin Research Institute Transgenic Facility was established in response to the need for transgenic and reproductive technologies by Institute investigators. Today, it offers a variety of services to investigators at McLaughlin Research Institute and from other institutions. This facility provides services necessary for investigators to create new mouse models for disease as well as models for testing gene function. Additional services include cryopreservation of mouse embryos and sperm, and rederivation of mouse lines to specific pathogen free status.
Facility Director, Dr. Teresa Gunn, works closely with requesting scientists to fulfill services as quickly and as efficiently as possible.
-
CRISPR/Cas9-mediated gene editing (experimental design service also offered)
Pronuclear Injection
ES Cell Injection
In Vitro Fertilization
Rederivation of mouse lines to specific pathogen free status
Embryo cryopreservation and recovery
Sperm cryopreservation and recovery
Please contact Dr. Gunn, using the form below, for service pricing.
Animal Resource Center
-
The MRI Animal Resource Center is a state-of-the-art facility, designed specifically to house mice in a safe and clean environment. The ARC facility was designed and constructed to meet exacting regulatory standards required for the operation of research animal facilities. The ARC has been fully AAALAC accredited since 1996.
McLaughlin Research Institute staff includes AALAS-certified Animal Care Technicians, Certified Veterinary Technicians, as well as skilled Cage Wash Technicians.
MRI’s Animal Resource Center currently has excess capacity over current faculty needs that is now available to help meet the demand for biomedical discovery and transgenic mouse services.
-
Provide healthy, genetically defined, specific-pathogen-free mice for use in IACUC-approved research, educational and testing protocols.
Ensure that research mice are cared for in a humane and appropriate manner by the provision of modern, well-maintained facilities, trained personnel, technical support, veterinary care and monitoring of animal care and use.
Work with Institute scientists and collaborators to ensure the availability of resources and technical assistance for their research projects.
Ensure that only pathogen-free mice are introduced into the facility by maintaining an active quarantine program and by working with the transgenic mouse facility to re-derive mice from other organizations that test positive for mouse pathogens.
Maintain an active surveillance and sentinel program for mouse pathogens.
Ensure observance of ethical standards and federal regulations pertaining to the care and use of mice for research, education and testing at McLaughlin Research Institute.
-
Rose Pitstick
Animal Resource Facility Director
rose@mclaughlinresearch.org

HERO:
Help Expand Research Opportunities
HERO Registry
The Challenge
Montana has the 6th oldest population ratio in the nation, with 18.5% of our total population over the age of 65. Given that the risk of development of neurodegenerative diseases increases with age, promoting the expansion of science and medicine via clinical research that targets advancements in diagnosis, treatment, and prevention of neurodegenerative diseases is a priority concern in our state.
The Need
The need for clinical research expansion in our state is vital. Montana has consistently ranked in the bottom 10% when it comes to participation and representation in clinical research in the United States. Of the clinical research that has been done in Montana, the vast majority of studies have focused on cancer research, with only 7.6% of total studies over time focusing on neurological disorders, and a meager 0.3% aimed at Alzheimer’s disease which, according to 2020 data from National Center for Health Statistics, is the 7th leading cause of death both nationally and locally in Montana.
The Solution
Scientists at the Institute are spearheading this patient registry which allows the Institute to enroll Montanans in potential clinical trials and research studies. They will use the registry to enroll individuals with mild cognitive impairment, Alzheimer’s, Parkinson’s, ALS and age-related macular degeneration, as well as individuals with no chronic disease as studies may seek healthy control participants.
Contract Research
-
McLaughlin Research Institute (MRI) has been widely recognized for its significant contributions to biomedical research especially in the generation of preclinical animal models and mouse genetics since 1954. MRI’s Animal Resource Center currently has excess capacity over current faculty needs that is now available to help meet the demand for biomedical discovery and transgenic mouse services. As a small, independent, mouse-only facility, the Institute is able to offer specific pathogen-free, secure mouse space and genetic expertise at a reasonable cost.
-
Historically, pharmaceutical companies have sought out MRI as a collaborator to accelerate drug target discovery in a cost-efficient manner by taking advantage of our first-rate facility, equipment, and expertise. Past and current for-profit collaborators include EMD Serono, Amgen, Celltech, Ionis Pharmaceuticals and Takeda Oncology (f/k/a Millenium Pharmaceuticals). MRI continues to foster collaborations with pharma although names of companies cannot always be disclosed. MRI currently has multiple collaborations with for-profit companies and other non-profit organizations to test therapeutics in pre-clinical mouse trials. MRI also has collaborations with academic researchers at Stanford University, Phenome Health, Inc, Touro University, Western Washington University, the University of Washington, Rutgers University, Columbia University, Harvard University, Wake Forest University, the University of Montana, and Montana State University who have taken the opportunity to expand their research at an affordable cost by using McLaughlin’s Animal Resource Center.
The user-friendly and service-oriented mouse facility at MRI features outstanding animal care and houses MRI’s transgenic facility for transgenic, reproductive, and cryopreservation technologies. Since 1996, our Animal Resource Center has been fully accredited by AAALAC International. The Institute’s skilled, AALAS-certified animal technicians adhere to rigorous animal health and genetic quality control practices. MRI enjoys the counsel of a strong Scientific Advisory Committee (SAC). Current SAC members are: Irving L. Weissman, MD (Chair), Stanford University School of Medicine; Leroy E. Hood, MD, PhD, Institute for Systems Biology; Frances Lefcort, PhD, Montana State University; Frank Bennett, Ionis Pharmaceuticals and N-Lorem and David Anderson, PhD, CalTech.
We also welcome traditional, investigator-initiated collaborations with MRI faculty: Deborah Cabin, mouse models for Parkinson’s disease and alpha-synuclein function; Teresa Gunn, mutations causing neurodegenerative disease; Tiffany Hensley-McBain; Mikael Klingeborn, role of exosomes in macular degeneration, protein misfolding, exosomal biomarkers, and therapeutics; Moses Leavens, protein biochemistry, biophysics, virology, and neurodegeneration; and Renee Reijo-Pera, Parkinson’s disease, human development and cell fate decisions.
-
1. Eichwald E (1956) The mite of immunology. Cancer Research 16, 918-20.
2. Eichwald E, Silmser C & Wheeler N (1957) The genetics of skin grafting. Ann NY Acad Sci 64, 737-740.
3. Eichwald E, Lustgraff E, Weissman I & Strainer M (1958) Attempts to demonstrate sex-linked histocompatibility genes. Transplant Bull 5, 387-388.
4. Eichwald E, Silmser C & Weissman I (1958) J Natl Cancer Inst 20, 563-575.
5. Dorf M, Stimpfling J & Benacerraf B (1979) Gene dose effects in Ir gene-controlled systems. J Immunol 123, 269-271.
6. Eichwald E (1988) Jack Stimpfling - The Voice from the Wilderness. Intl Rev Immunol 3, 281-283.
7. Bermingham JR, Shearin H, Pennington J, O'Moore J, Jaegle M, Driegen S, van Zon A, Darbas A, Özkaynak E, Ryu EJ, Milbrandt J & Meijer D (2006) The claw paw mutation reveals a role for Lgi4 in peripheral nerve development. Nature Neurosci 9, 76-84.
8. Henson V, Palmer L, Banks S, Nadeau JH & Carlson GA (1991) Loss of heterozygosity and mitotic linkage maps in the mouse. Proc Natl Acad Sci 88, 6486-6490.
9. Zou D, Silvius D, Fritzsch B & Xu P-X (2004) Eya1 and Six1 are essential for early steps of sensory neurogenesis in mammalian cranial placodes Development 131, 5561-5572.
10. Gnanaprakash M, Staniszewski A, Zhang H, Pitstick R, Kavanaugh MP, Arancio O & Nicholls RE (2021) Leucine carboxyl methyltransferase 1 overexpression protects against cognitive and electrophysiological impairments in Tg2576 APP transgenic mice. J Alzheimers Dis 79, 1813-1829.
11. Sotelo J, Canclini L, Kun A, Sotelo‐Silveira J, Calliari A, Cal K, Bresque M, DiPaolo A, Farias J & Mercer J (2014) Glia to axon RNA transfer. Dev Neurobiol 74, 292-302.
12. Li F, Calingasan N, Yu F, Mauck W, Toidze M, Almeida C, Takahashi R, Carlson G, Beal F, Lin M & Gouras G (2004) Increased plaque burden in brains of APP mutant MnSOD heterozygous knockout mice. J Neurochem 89, 1308-1312.
13. Wang K, Lee I, Carlson G, Hood L & Galas D (2010) Systems biology and the discovery of diagnostic biomarkers. Disease Markers 28, 199-207.
14. Kopeikina K, Polydoro M, Tai H-C, Yaeger E, Carlson G, Pitstick R, Hyman B & Spires‐Jones T (2013) Synaptic alterations in the rTg4510 mouse model of tauopathy. J Comp Neurol 521.
15. Stephenson D, Chiotti K, Ebeling C, Groth D, DeArmond S, Prusiner S & Carlson G (2000) Quantitative trait loci affecting prion incubation time in mice. Genomics 69, 47-53.
16. Fanara P, Wong PY, Husted KH, Liu S, Liu VM, Kohlstaedt LA, Riiff T, Protasio JC, Boban D, Killion S, Killian M, Epling L, Sinclair E, Peterson J, Price RW et al. (2012) Cerebrospinal fluid-based kinetic biomarkers of axonal transport in monitoring neurodegeneration. J Clin Invest 122, 3159-3169.
17. Arand J, Chiang H, Martin D, Snyder M, Sage J, Reijo Pera RA & Wossidlo M (2021) Tet enzymes are essential for early embryogenesis and completion of embryonic genome activation. EMBO Reports e53968.
18. Ament SA, Pearl JR, Grindeland A, St Claire J, Earls JC, Kovalenko M, Gillis T, Mysore J, Gusella JF, Lee JM, Kwak S, Howland D, Lee MY, Baxter D, Scherler K et al. (2017) High resolution time-course mapping of early transcriptomic, molecular and cellular phenotypes in Huntington's disease CAG knock-in mice across multiple genetic backgrounds. Hum Mol Genet 26, 913-922.
19. George L, Chaverra M, Wolfe L, Thorne J, Close-Davis M, Eibs A, Riojas V, Grindeland A, Orr M, Carlson G & Lefcort F (2013) Familial dysautonomia model reveals Ikbkap deletion causes apoptosis of Pax3+ progenitors and peripheral neurons. Proc Natl Acad Sci 110, 18698-18703.
20. Chaverra M, George L, Mergy M, Waller H, Kujawa K, Murnion C, Sharples E, Thorne J, Podgajny N, Grindeland A, Ueki Y, Eiger S, Cusick C, Babcock A, GA GC et al. (2017) The familial dysautonomia disease gene IKBKAP is required in the developing and adult mouse central nervous system. Dis Model Mech 10, 605-618.
21. Minikel EV, Zhao HT, Le J, O'Moore J, Pitstick R, Graffam S, Carlson GA, Kavanaugh MP, Kriz J, Kim JB, Ma J, Wille H, Aiken J, McKenzie D, Doh-Ura K et al. (2020) Prion protein lowering is a disease-modifying therapy across prion disease stages, strains and endpoints. Nucleic Acids Res 48, 10615-10631.
22. Vallabh S, Zou D, Pitstick R, O'Moore J, Peters J, Silvius D, Kriz J, Jackson W, Carlson G, Minikel E & Cabin D (2022) Therapeutic trial of anle138b in mouse models of genetic prion disease. J Virology JVI01672-22R1 (in press).
23. Kong J, Young C, Pusapati G, Patel C, Ho S, Krishnan A, Lin J-I, Devine W, de Bellaing AM, Athni T, Aravind L, Gunn T, Lo C & Rohatgi R (2020) A ubiquitin-based mechanism for the oligogenic inheritance of heterotaxy and heart defects. bioRxiv doi.org/10.1101/2020.05.25.113944.
24. Kong JH, Young CB, Pusapati GV, Espinoza FH, Patel CB, Beckert F, Ho S, Patel BB, Gabriel GC, Aravind L, Bazan JF, Gunn TM, Lo CW & Rohatgi R (2021) Gene-teratogen interactions influence the penetrance of birth defects by altering Hedgehog signaling strength. Development 148.
25. Gunn TM & Carlson GA (2013) RML prions act through Mahogunin and Attractin-independent pathways. Prion 7, 267-271.
26. Cui J, Carey J & Reijo Pera RA (2022) Identification of DOT1L inhibitor in a screen for factors that promote dopaminergic neuron survival. Front Aging Neurosci 14, 1026468.
27. Anderson S, Lee I, Ebeling C, Stephenson D, Schweitzer K, Baxte D & Gunn T (2015 ) Disrupted SOX10 function causes spongiform neurodegeneration in gray tremor mice. Mammalian Genome 26, 80-93.
28. Mortberg M, Zhao H, Reidenbach A, Gentile J, Kuhn E, O'Moore J, Dooley P, Mazur C, Allen S, Trombetta B & McManus A (2021) Regional variability, genotypic effects, and pharmacodynamic impact on prion protein concentration in the central nervous system. medRxiv Jan 1.